Best Endovascular vs. Best Surgical Therapy in Patients with Critical Limb Ischemia (BEST-CLI)
About BEST-CLI
BEST-CLI is an international research study aimed at figuring out the best treatment for people with peripheral arterial disease (PAD) - also known as poor blood flow, and associated with Critical Limb Ischemia (CLI). It is Federally funded by the National Heart, Lung and Blood Institute at the National Institutes of Health.
November 2022 update: BEST-CLI enrollment ended in October of 2019, and completed a minimum of 24 months’ follow-up for all patients in October of 2021. The primary clinical and quality of life results will be presented at the American Heart Association in Chicago, Illinois on Monday, November 7.
What is peripheral arterial disease and who is at risk?
Peripheral arterial disease (PAD) is a condition in which plaque builds up in the blood vessels that carry blood to arms and legs. About 3-10% of people have PAD and this number increases to 15-20% in those over 70 years of age. PAD is particularly common in individuals who smoke or have diabetes. Some patients with PAD develop critical limb ischemia (CLI). In CLI, blood vessels that deliver blood to the leg and foot are so narrowed by plaque buildup (atherosclerosis) that the foot cannot get enough blood. CLI can cause foot pain at rest, foot and leg ulcerations, and lead to gangrene and loss of the leg.
How is peripheral arterial disease treated?
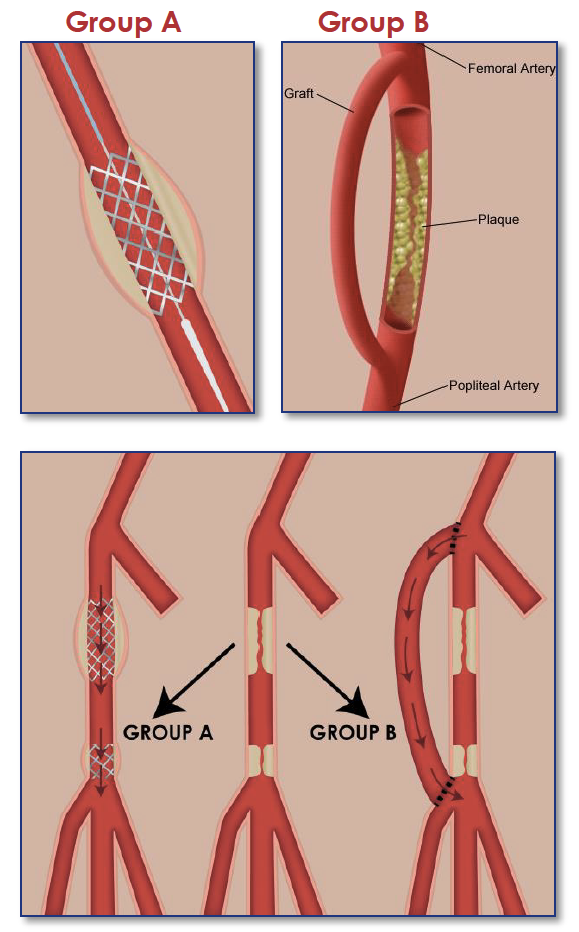
CLI is typically treated with revascularization procedures (creating detours around or through the narrowed or blocked arteries) to improve blood flow to the leg and foot. There are two types of revascularization procedures that can be used to treat the CLI: open surgery and endovascular treatment.
Open Surgery
During this surgery, a doctor takes your own leg vein and connects it to a healthy artery above the blockages and then to an artery in your leg below the blockages. The grafted vein goes around (bypasses) the blocked part of the artery. This bypass provides a new pathway for blood to your leg and foot. Some people do not have a leg vein that can be used. In these individuals, an artificial graft is used.
Endovascular Treatment
During this procedure, a thin flexible tube (catheter) is inserted through an artery in the groin or arm and carefully guided into the artery that is narrowed. Once the tube reaches the narrowed artery, a small balloon at the end of the tube is inflated. The balloon may remain inflated from 20 seconds to 3 minutes. The pressure from the inflated balloon presses fat and calcium deposits (plaque) against the wall of the artery to improve blood flow. Once the fat and calcium buildup is compressed, a small, expandable wire-mesh tube called a stent is sometimes inserted into the artery to hold it open. The use of stents has not been studied specifically in patients with critical limb ischemia, and is therefore considered investigational (new). Plaque removing devices may also be used to remove plaque in the blockages.
What happened in the study?
Patients interested in participating in the BEST-CLI study were asked to come to a local BEST-CLI study site for further tests, to answer questions about your health, and so that they may learn more about the study.
Those who chose to participate were be assigned by chance (as in the flip of a coin) to receive either surgery or the endovascular procedure. After the procedure, patients were asked to return to your BEST-CLI physician for follow-up exams at 1, 3, 6, 12, 18, and 24 months, and annually after that.
Patients also received phone calls from the study nurse at 30, 42, 54, 66 and 78 months.
There are 150+ medical centers that participated in this study. BEST-CLI targeted enrollment of 2,100 participants across the United States, Canada, Italy, Finland and New Zealand. The physicians chosen to participate in BEST-CLI were selected because they have previous experience and are skilled at performing the procedures.
Your doctor can tell you more about this study and answer any questions you may have. Patients who agreed to take part in the study were asked to sign a consent form. Your doctor will then tell you whether or not you might have been eligible to participate based on your clinical condition.
All information collected throughout this study remains strictly confidential. Patient name and records will not be given out without the patient's' consent.
References
1. Norgren L, Hiatt WR, Dormandy JA, Nehler MR, Harris KA, Fowkes FG. Inter-Society Consensus for the Management of Peripheral Arterial Disease (TASC II). J Vasc Surg. Jan 2007;45 Suppl S:S5-67.
2. Selvin E, Erlinger TP. Prevalence of and risk factors for peripheral arterial disease in the United States: results from the National Health and Nutrition Examination Survey, 1999-2000. Circulation. Aug 10 2004;110(6):738-743.
Frequently Asked Questions
Are these treatments new or experimental?
No. Endovascular therapy and bypass surgery are two standard types of treatment that improve blood flow to the leg. This study is comparing these two standard-of-care treatments.
What will happen during this study?
Participants in this study were randomly assigned to one of two treatment groups: either endovascular therapy (group A) or surgical therapy (group B).
If you were assigned to Group A, the blockage in your artery was opened with a ballon and a wire mesh stent may be inserted permanently to maintain the opening.
If you were assigned to Group B, a detour or "bypass" around the blockage in your artery was made by inserting a vein or tube made of synthetic material.
Who is eligible to partipate?
Patients were eligible if they had:
Symptoms such as leg pain, gangrene, or foot ulcers
Blockage of one or more of the blood vessels located below your groin
And the BEST-CLI investigator confirmed you were eligible for both endovascular or surgical treatment
What are my costs in this study?
Depending on your health insurance, patients may have been required to make co-payments for visits and tests that are considered standard care for CLI. All tests, visits, and procedures that are done only for research purposes were paid for by NHLBI.
Will I be paid for participation in this study?
Patients received $50.00 upon successful completion of each study visit. (Participants with unusual travel burdens may have received up to $100.00 per visit.)
How will patient safety be monitored?
In addition to your study physician's commitment to safety, an independent Data and Safety Monitoring Board (DSMB) regularly reviewed study data and make recommendations to the National Heart, Lung and Blood Institute (NHLBI) regarding patient safety in this trial.
What are the risks?
The risks are the same as those you would face if undergoing either of the two standard treatments for CLI. The risks include limb (leg) complications and systemic (whole body) complications.
What are the benefits?
Participation in the study helped to gather information about these CLI treatments. This information will help doctors and patients make informed treatment decisions for future CLI patients. Patients themselves may not have benefitted personally from being in this research study.
Where are the study locations?
Physicians around the world specialize in treating CLI. These doctors, research nurses, and coordinators on "BEST-CLI teams" provide high-quality, long-term care to patients who enroll in this study.
There were over 150 BEST-CLI study locations with nearly 1000 participating physicians in the US, Canada, Finland, Italy and New Zealand.
Clinicaltrials.gov has up-to-date information on the current status of this trial.